A) Nonpolar
B) Covalent
C) Polar covalent
D) Ionic
F) A) and B)
Correct Answer

verified
Correct Answer
verified
True/False
The alkane solvents are quite soluble in water.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What do neurotransmitters do?
A) Travel outside the neuron, cross a small gap, and reenter the neuron
B) Travel through the neuron, cross a small gap, and arrive at the other side of the neuron
C) Travel outside the neuron, cross a small gap, and arrive at the next neuron
D) Travel through the neuron, cross a small gap, and reenter the neuron
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which is the correct Lewis dot structure for chlorine?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The following molecule, phenol, is used in some medicines. How many carbon atoms in it have a tetrahedral geometry?
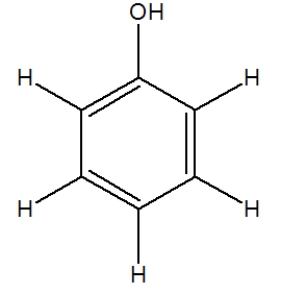
A) 0
B) 1
C) 2
D) 3
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How do many illegal drugs cause a "high"?
A) By halting brain activity
B) By altering the biochemical processes that occur in the brain
C) By altering the biochemical processes that occur in the brain and other vital organs
D) By altering the body's temperature-regulating mechanisms
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the correct name for the illegal drug "meth"?
A) d-methyleneamphetamine
B) d-methamphetamine
C) l-methamphetamine
D) l-methyleneamphetamine
F) C) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
How do neurons communicate with each other?
Correct Answer

verified
By sending an electr...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which is the correct Lewis dot structure for He?
A) He:
B) H:
C) He.
D) H.
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How does Lewis theory explain the loss or gain of electrons in an ionic bond?
A) Atoms are attempting to gain electrons.
B) Atoms are attempting to lose electrons.
C) Atoms are attempting to share valence electrons.
D) Atoms are attempting to achieve a noble gas electron configuration.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the term for two molecules with the same formula but two different structures?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following would be correct structure for the nitrite ion, NO2- ?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Lewis structures with single lines indicate what kind of bonding?
A) Covalent, 2-electron bonding
B) Ionic, 2-electron bonding
C) Covalent, 4-electron bonding
D) Ionic, 4-electron bonding
F) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following represent the best structure for SO2?
A) ![]()
B) ![]()
C) ![]()
D) ![]()
F) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What kind of bond connects the carbon atom and oxygen atom in carbon monoxide?
A) A single bond
B) A double bond
C) A triple bond
D) Two single bonds
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What does the term "resonance structure" mean?
Correct Answer

verified
Two or more Lewis structures, ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
How many carbons in the following molecule have a tetrahedral geometry?
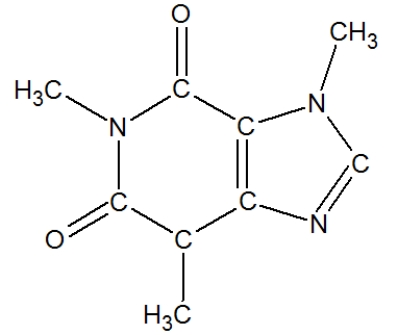
A) 2
B) 3
C) 4
D) 5
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Short Answer
What is the proper term for the nerve cells that make up the human nervous system?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What do the Lewis structures of all the alkali metals have in common?
A) A complete sub-shell of electrons
B) One outer electron
C) Two outer electrons
D) Three outer electrons
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the electronegativity difference in the C-O bond?
A) 1.0
B) 2.5
C) 3.5
D) 0
F) A) and C)
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 123
Related Exams